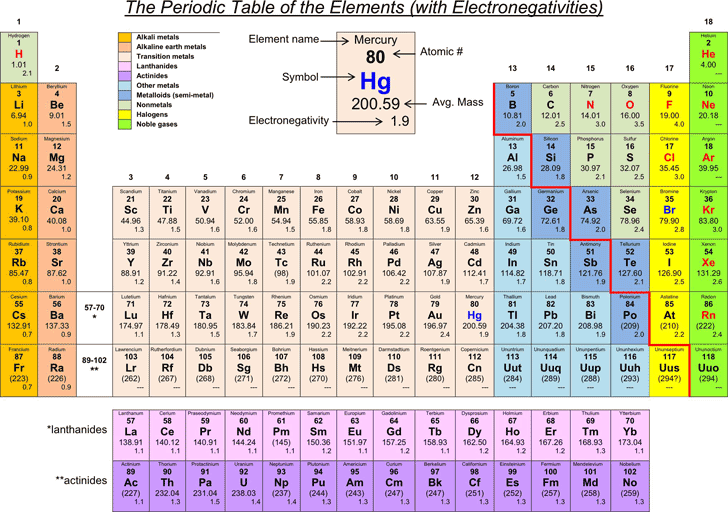
Standard temperature is equal to 0☌ or 32☏ and standard pressure is equal to 1 atm, 101.3kPa or 760 mmHg (torr)ĥ) Electronegativity given by the Pauling scale. Temperature converterģ) Boiling point is the temperature at which a substance changes from liquid to gas state.Ĥ) Density (mass/volume) at standard temperature and pressure, given as gram/cm 3. u = atomic mass unit, 1 u = 1.66 10 -27 kgĢ) Melting point is the temperature at which a substance changes from solid to liquid state.As you can see, electronegativities generally increase from left to right across a period and decrease down a group.Elements of the Periodic System - Physical Properties Element Nameġ) Standard average atomic weights (IUPAC 1997) for the isotops naturally present in the element. A value of 4.0 is assigned to fluorine, the most electronegative element. The periodic table below shows the Pauling electronegativity scale. Electronegativity is a measure of an atoms ability to attract shared electrons to itself. Than the other atom, the electrons will not be shared and an ionic bond will If one atom is overwhelmingly more electronegative If one atom is more electronegative, the electrons of the bond are Two atoms of the bond are of equal electronegativity, the electrons are equally A high electronegativity value means an atom readily attracts electrons to form a chemical bond with another atom. Electronegativity is a measure of how easily an atom attracts a pair of electrons to form a chemical bond. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6. Periodic table showing the trend from low to high of the element’s electronegativity. Team is no longer able to hold onto the rope and the entire rope ends up on The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. If one team is overwhelmingly stronger, the weaker Image showing periodicity of the chemical elements for electronegativity (Sanderson) in a periodic table heatscape style. The ground state electronic configuration of neutral radon is Xe. Image showing periodicity of the chemical elements for electronegativity (Sanderson) in a periodic table cityscape style. Radon atoms have 86 electrons and the shell structure is 2.8.18.32.18.8. If one team is stronger, the rope is pulled Electronegativity (Sanderson) Gallery of images. Will not be shared at all the more electronegative atom will "take" them resulting If the difference in electronegativity is large enough, the electrons Of the atoms (because it is more electronegative), the electrons will be unequally Electronegativity, symbol, is a chemical property that describes the tendency of an atom to attract electrons towards this atom. If the electrons of a bond are more attracted to one Therefore the electronegativity is greatest at the top-right of the periodic table and decreases toward the bottom-left. 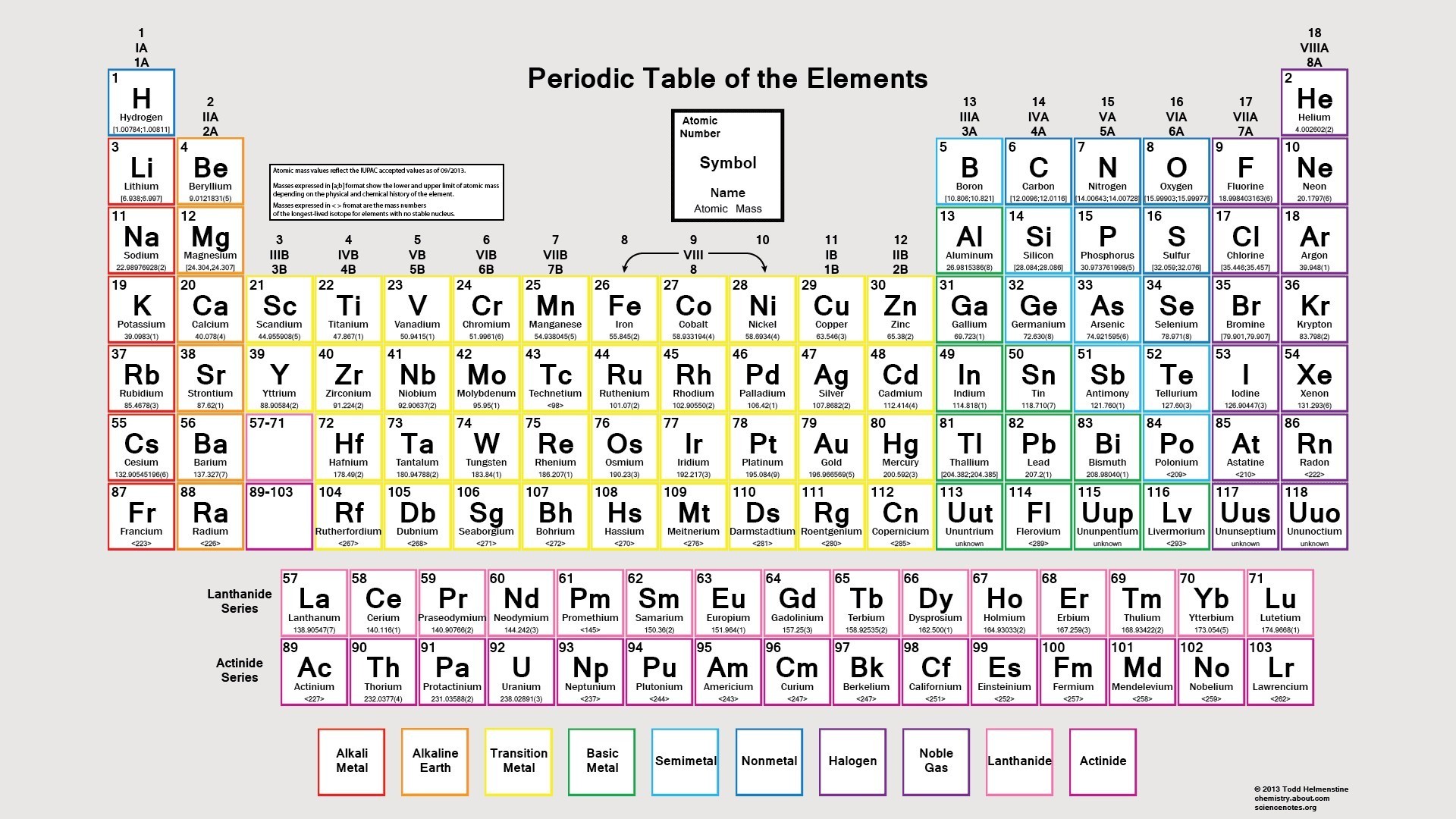
If atoms bonded together have the same electronegativity, the shared electrons 5s2 and the term symbol of technetium is 6S5/2. The ground state electronic configuration of neutral technetium is Kr. Technetium atoms have 43 electrons and the shell structure is 2.8.18.14.1. An atom's ability to attract the shared electrons of a covalent bond to itself. Group name: (none) Period in periodic table: 5.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
